Bleeding Risk with Apixaban vs. Rivaroxaban in Acute Venous Thromboembolism
Lana A. Castellucci, Vivien M. Chen, Michael J. Kovacs, et al
N Engl J Med 2026;394:1051-1060
DOI: 10.1056/NEJMoa2510703
Abstract
BACKGROUND
Apixaban and rivaroxaban are the oral anticoagulants most frequently used to treat acute venous thromboembolism. However, uncertainty remains about the difference in bleeding risk between the two medications.
METHODS
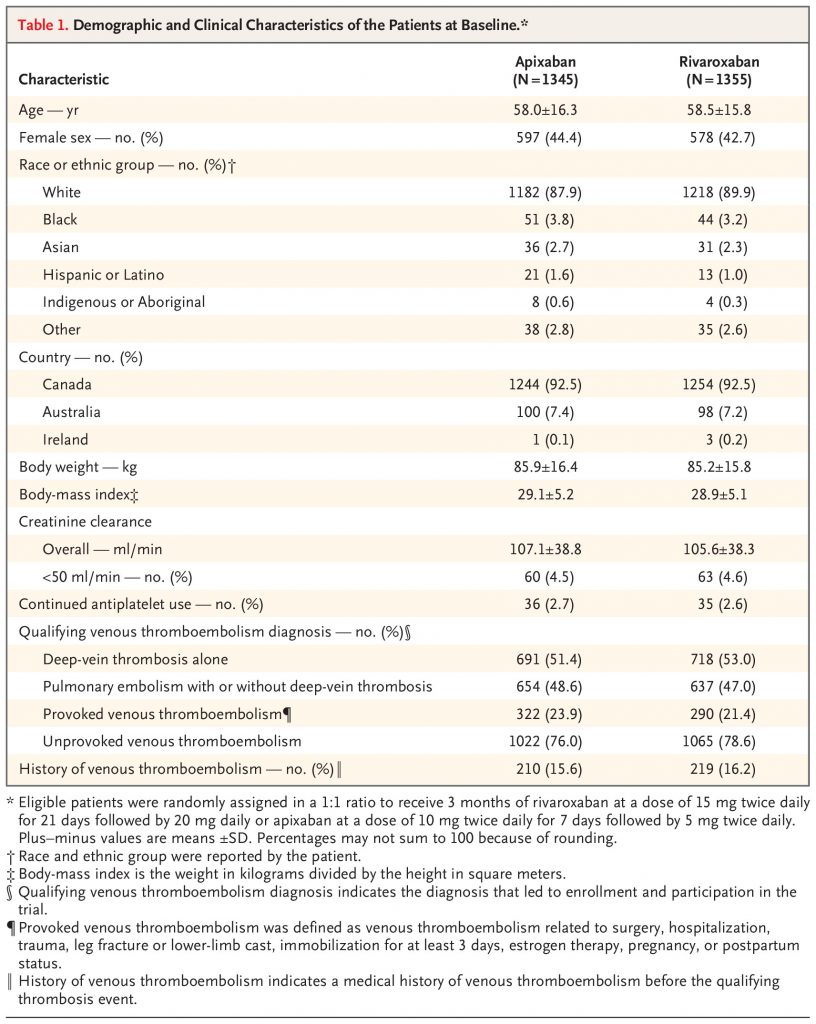
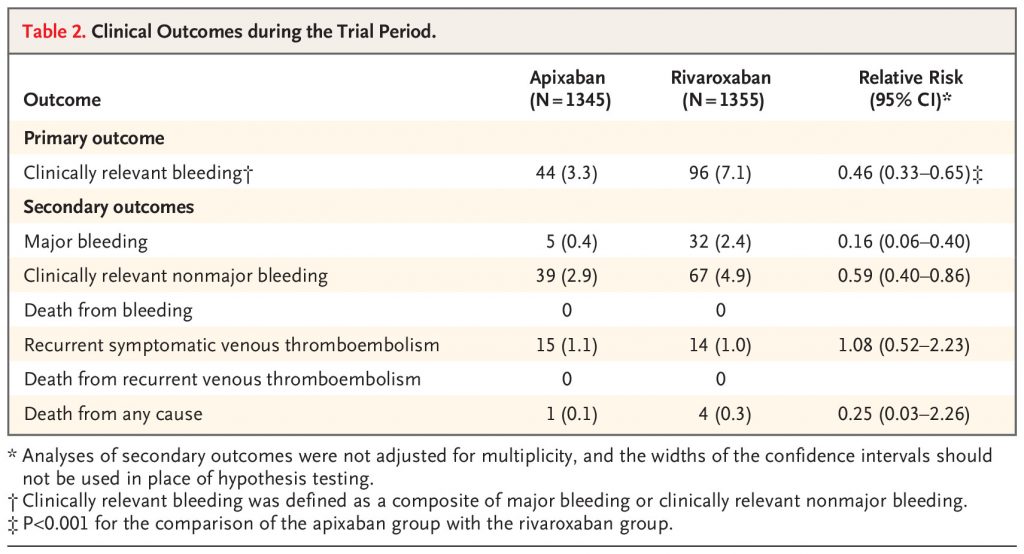
In an international trial with a prospective, randomized, open-label, blinded end-point design, we assigned, in a 1:1 ratio, eligible patients with acute symptomatic pulmonary embolism or proximal deep-vein thrombosis to receive apixaban or rivaroxaban for 3 months. Apixaban was given at a dose of 10 mg twice daily for 7 days followed by 5 mg twice daily, and rivaroxaban was given at a dose of 15 mg twice daily for 21 days followed by 20 mg daily. The primary outcome was clinically relevant bleeding, a composite of major bleeding or clinically relevant nonmajor bleeding, as defined according to the International Society on Thrombosis and Haemostasis, during the 3-month trial period. Secondary outcomes included death from any cause.
RESULTS
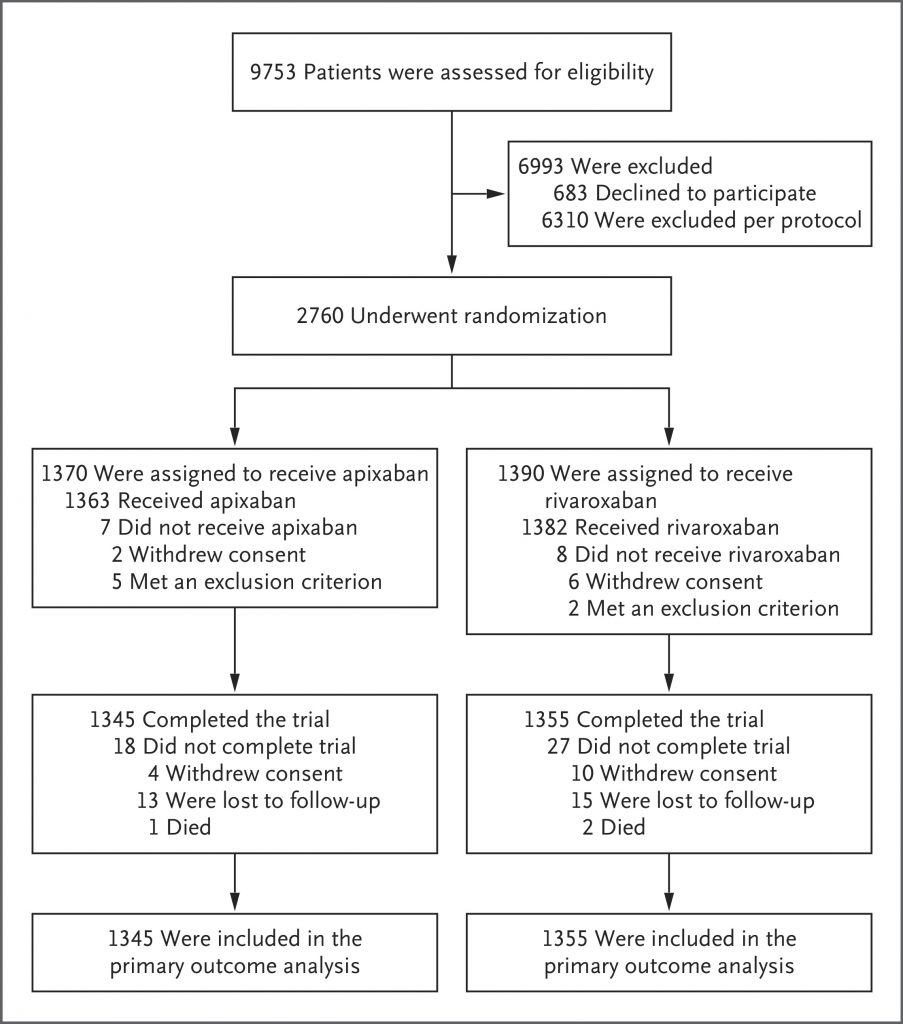
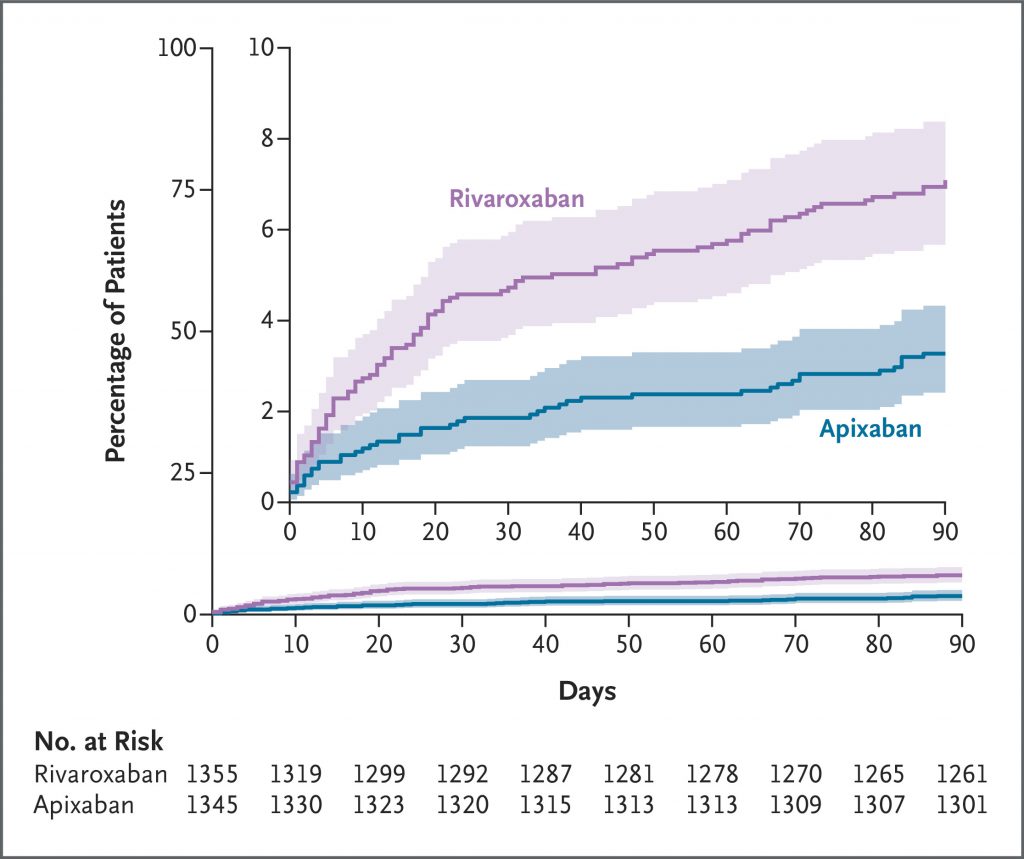
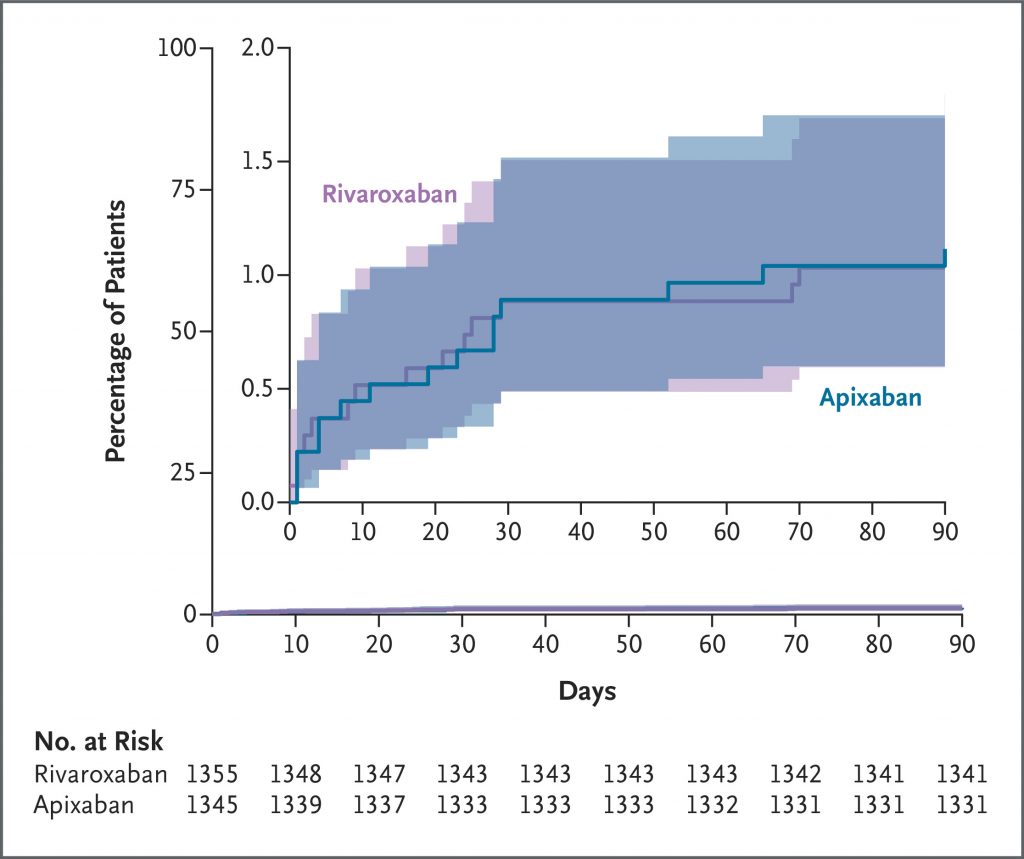
A total of 2760 patients underwent randomization: 1370 to the apixaban group and 1390 to the rivaroxaban group. A primary-outcome event occurred in 44 of 1345 patients (3.3%) in the apixaban group and 96 of 1355 patients (7.1%) in the rivaroxaban group (relative risk, 0.46; 95% confidence interval [CI], 0.33 to 0.65; P<0.001). Death from any cause occurred in 1 patient (0.1%) in the apixaban group and in 4 patients (0.3%) in the rivaroxaban group (relative risk, 0.25; 95% CI, 0.03 to 2.26). Serious adverse events unrelated to bleeding or venous thrombosis occurred in 36 patients (2.7%) in the apixaban group and in 30 patients (2.2%) in the rivaroxaban group.
CONCLUSIONS
Among patients with acute venous thromboembolism, the risk of clinically relevant bleeding was significantly lower with apixaban than with rivaroxaban during the 3-month treatment period. (Funded by the Canadian Institutes of Health Research and others; COBRRA ClinicalTrials.gov number, NCT03266783.)