JOURNAL ARTICLE EDITOR'S CHOICE
Retreatment With Nirmatrelvir/Ritonavir Following Return of COVID-19 Symptoms and SARS-CoV-2 Positivity
Wuyan Zhang, Steven G Terra, Edward Alan Weinstein, et al
Clinical Infectious Diseases, Volume 82, Issue 1, 15 January 2026, Pages e33–e40, https://doi.org/10.1093/cid/ciaf548
Abstract
Background
Rebound of coronavirus disease 2019 (COVID-19) is defined as return of symptoms and/or viral positivity after resolution of the initial infection. While rebound is typically mildly symptomatic and rarely associated with progression to severe disease, the benefit of repeated antiviral treatment has not been investigated.
Methods
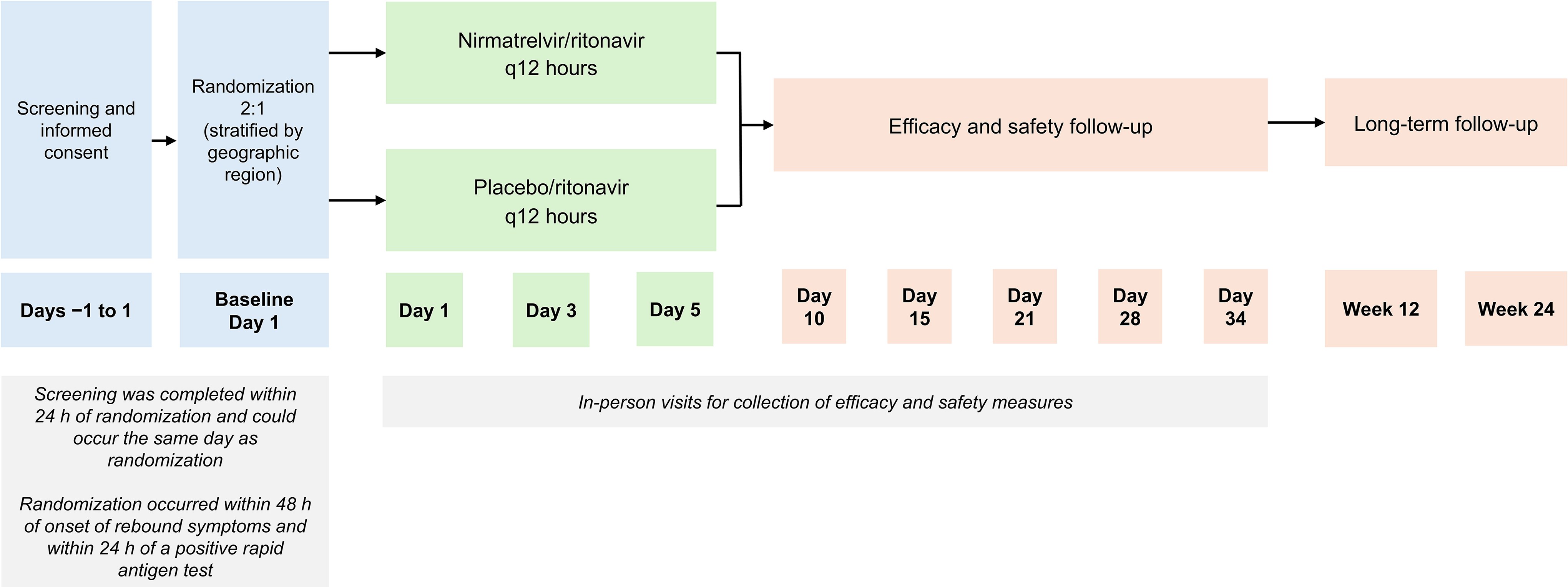
This randomized, double-blind, placebo-controlled clinical trial evaluated the efficacy and safety of a second 5-day treatment course of nirmatrelvir/ritonavir versus placebo/ritonavir in participants with symptomatic mild to moderate COVID-19 with a positive severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) rapid antigen test within 14 days of initial nirmatrelvir/ritonavir treatment. The primary efficacy endpoint was the change in viral SARS-CoV-2 RNA level from baseline to Day 5.
Results
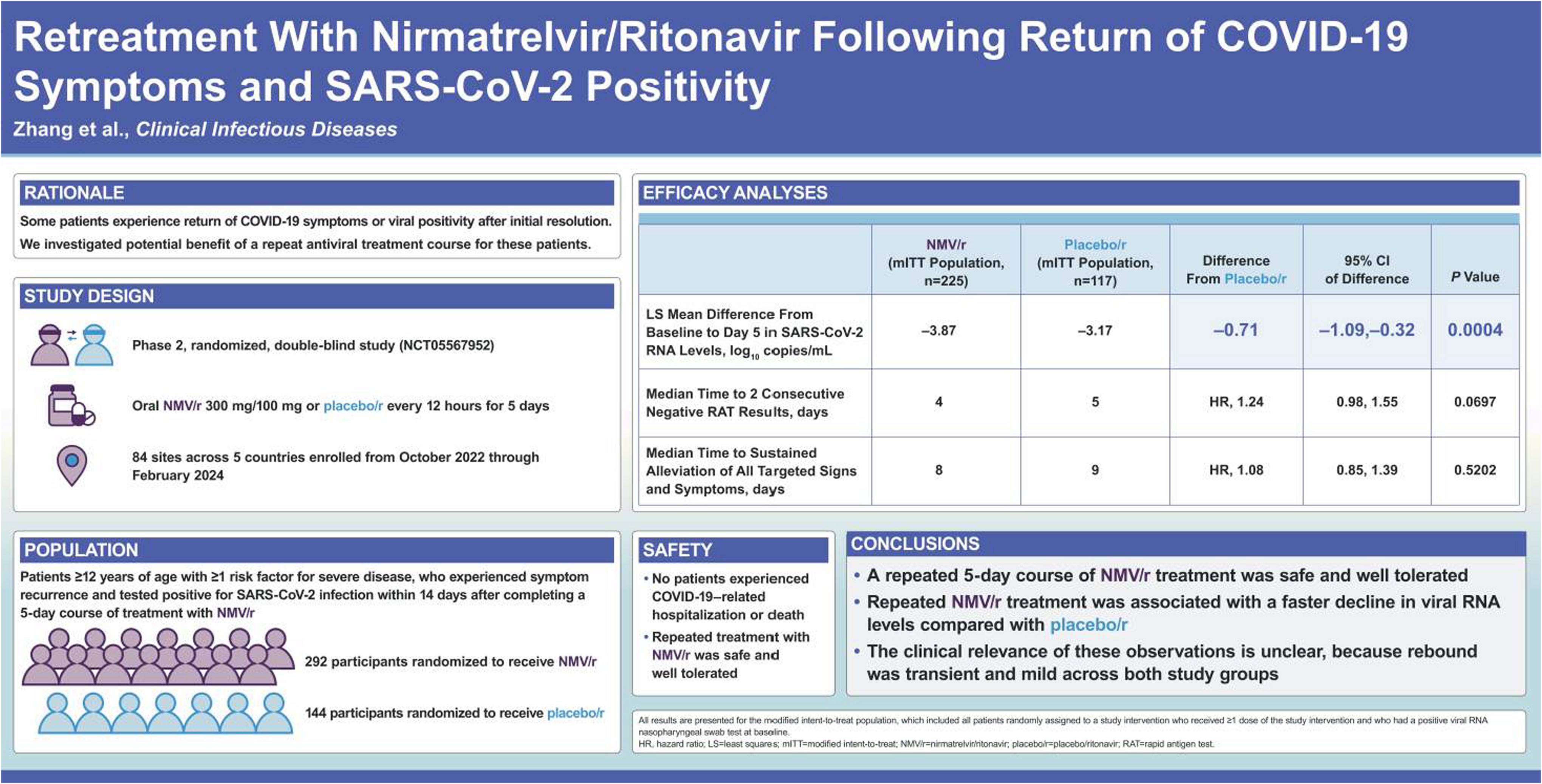
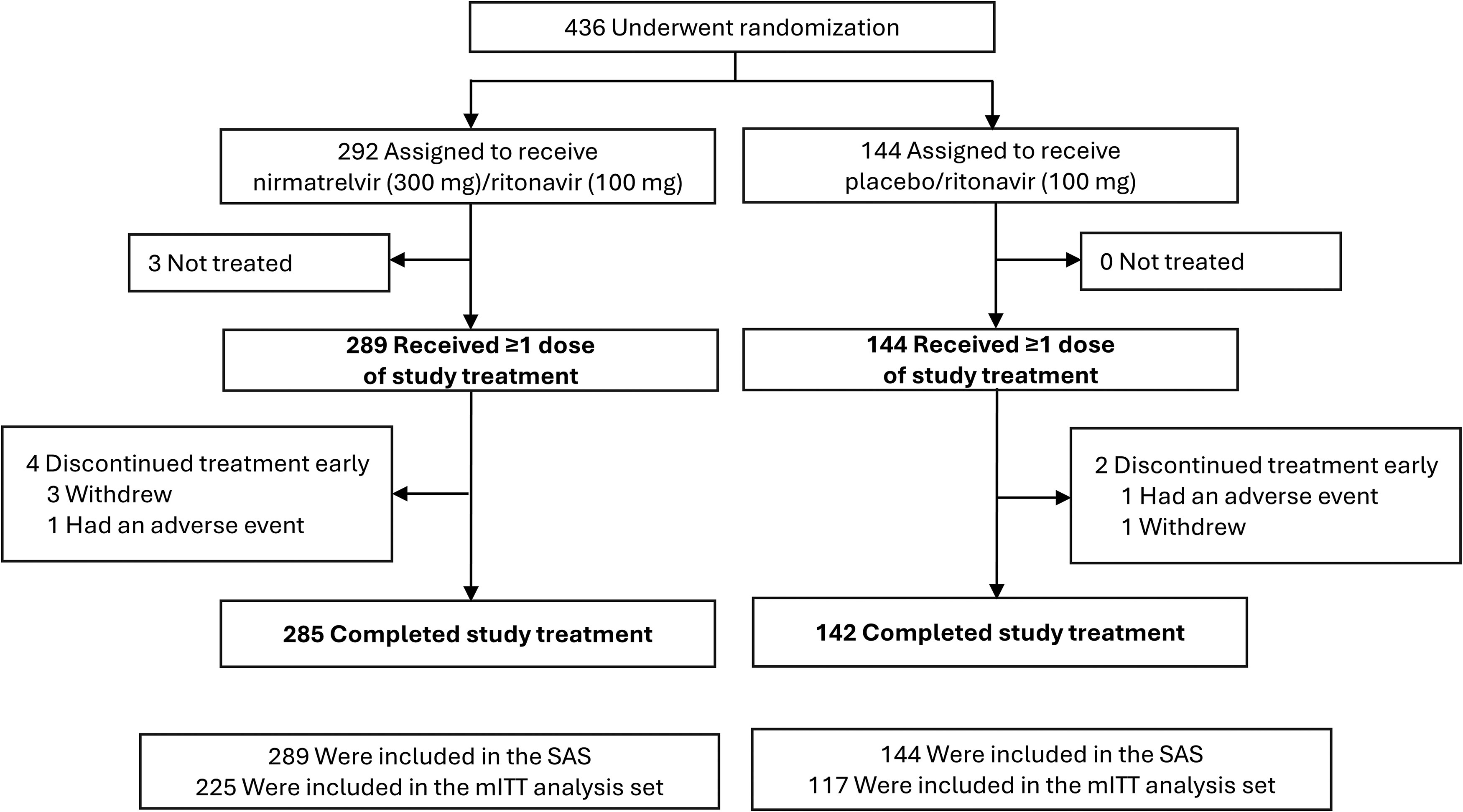
The full analysis set included 436 participants assigned to treatment (292, nirmatrelvir/ritonavir; 144, placebo/ritonavir). A second 5-day course of nirmatrelvir/ritonavir resulted in a significant reduction in viral RNA levels at Day 5 compared with placebo/ritonavir (P = .0004; 95% CI, −1.09, −.32). The median time to 2 consecutive negative RAT results was 4 versus 5 days, and the median time to sustained alleviation of all targeted signs/symptoms was 8 versus 9 days in the nirmatrelvir/ritonavir and placebo/ritonavir groups, respectively. Retreatment with nirmatrelvir/ritonavir was safe and well tolerated, and there were no occurrences of COVID-19–related hospitalizations or deaths.
Conclusions
A repeated 5-day course of nirmatrelvir/ritonavir was safe and well tolerated and was associated with a faster decline in viral RNA levels. In this study, there was no clear benefit of retreatment because rebound was transient and mild and did not lead to severe COVID-19.
Clinical Trials Registration
NCT05567952; https://clinicaltrials.gov/study/NCT05567952.