Acute Respiratory Distress Syndrome Molecular Phenotypes Have Distinct Lower Respiratory Tract Transcriptomes
Aartik Sarma, Stephanie A. Christenson, Beth Zha Shoshana, et al
Am J Respir Crit Care Med 2025; 211: 2352-2362
https://doi.org/10.1164/rccm.202407-1454OC
Abstract
Rationale: Two molecular phenotypes of the acute respiratory distress syndrome (ARDS) with divergent clinical trajectories and responses to therapy have been identified. Classification as “hyperinflammatory” or “hypoinflammatory” depends on plasma biomarker profiling. Limited data are available about the differences in the pulmonary biology of the molecular phenotypes.
Objectives: To identify differences in the pulmonary biology of ARDS molecular phenotypes
Methods: We compared tracheal aspirate gene expression between hyperinflammatory and hypoinflammatory phenotypes in bulk RNA sequencing (RNASeq) from coronavirus disease (COVID-19) and non–COVID-19 ARDS and single-cell RNASeq from non–COVID-19 ARDS. In a subset of subjects, we also compared plasma proteomic data.
Measurements and Main Results: In bulk RNASeq analyses, 1,157 genes were differentially expressed (false discovery rate < 0.1) between phenotypes in non–COVID-19 ARDS, and 85 genes were differentially expressed between phenotypes in COVID-19 ARDS. Eighteen genes were reproducibly differentially expressed between phenotypes in both cohorts, including greater expression of IL32, HSPA8, and PPP3CCin hyperinflammatory ARDS. A total of 195 pathways were reproducibly enriched across the two cohorts by gene set enrichment analysis, including greater expression of granulopoiesis, T-cell and IFN signaling, and integrated stress response pathways in hyperinflammatory ARDS. Network analysis of single-cell RNASeq in a third group of patients identified greater T-cell signaling to other immune cells in hyperinflammatory ARDS.
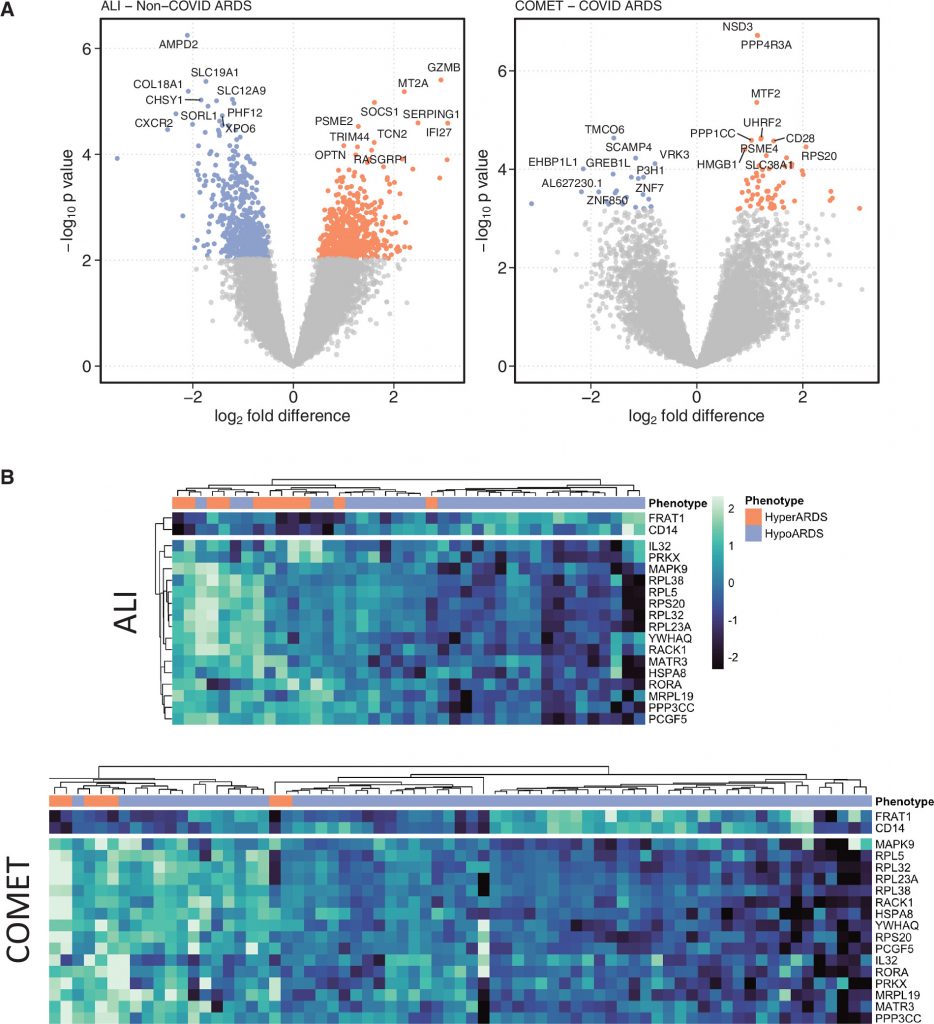
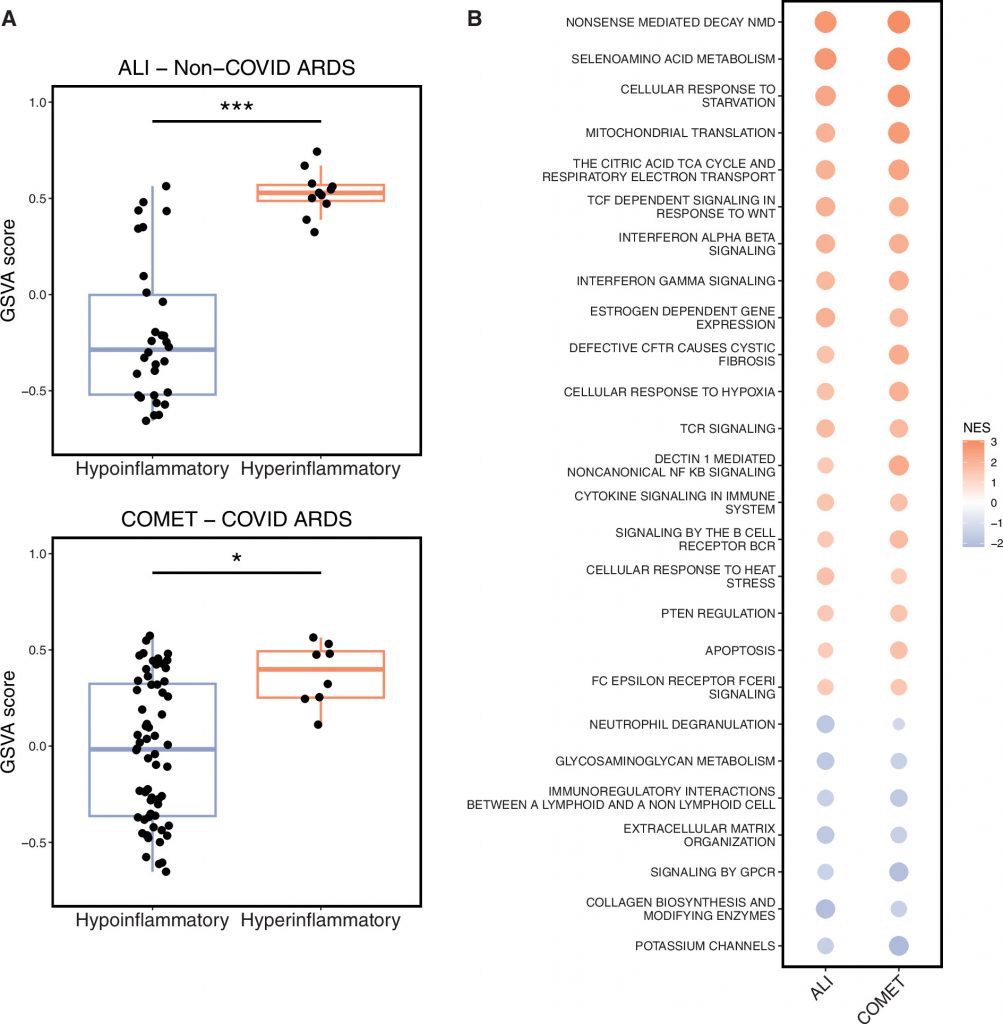
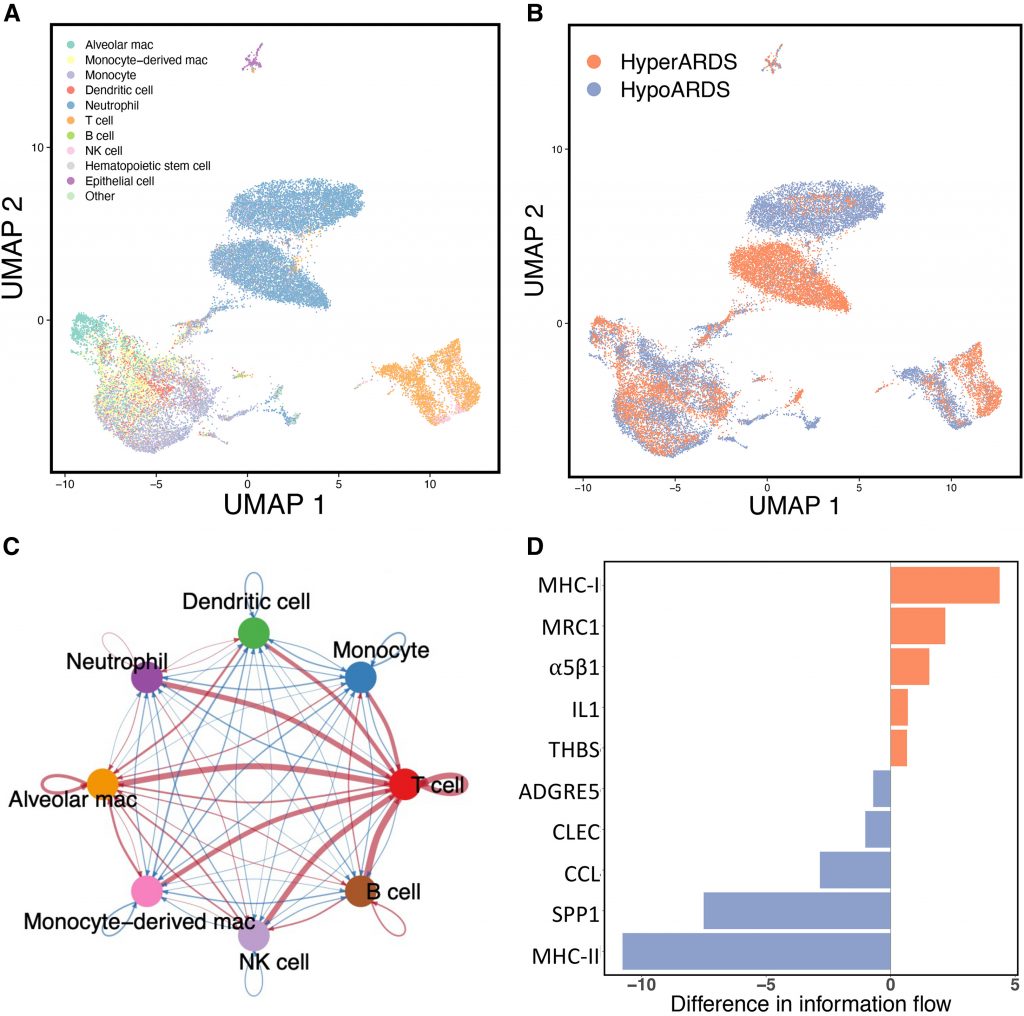
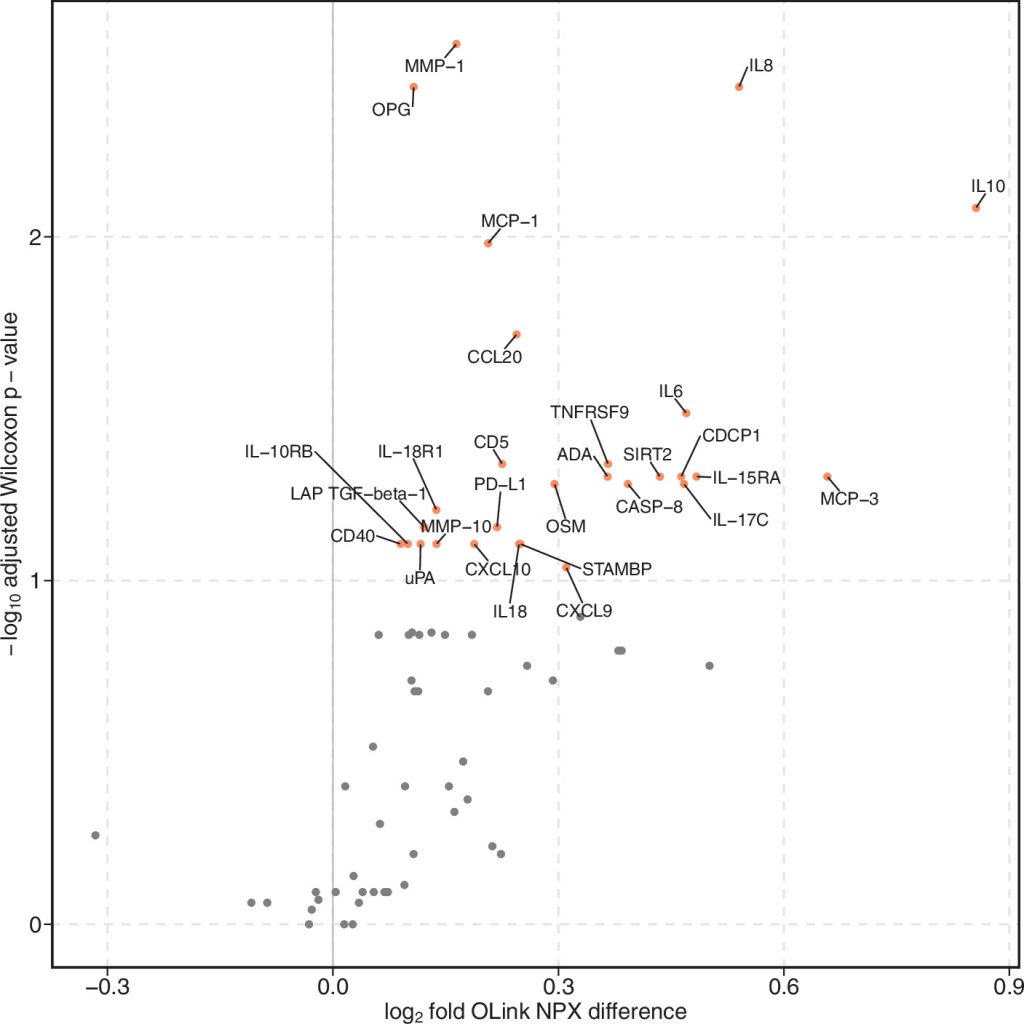
Conclusions: Hyperinflammatory and hypoinflammatory ARDS molecular phenotypes have distinct respiratory biology. Hyperinflammatory ARDS is characterized by an increased IFN-stimulated gene expression and T-cell activation in the lungs.